How To Draw Dash And Wedge Chemistry
i.7: 3D Structure
- Page ID
- 225766
Dashed-wedged line structure
A widely used way of showing the 3D structure of molecules is based on the skeletal formula, merely using of dashes, wedges, and straight lines. This drawing method is essential because the placement of different atoms could yield dissimilar molecules even if the molecular formulas were exactly the same. Below are 2 drawings of a iv-carbon molecule with two chlorines and two bromines attached.
Both drawings look like they represent the aforementioned molecule; however, if we add dashes and wedged bonds we volition encounter that ii dissimilar molecules could exist depicted:
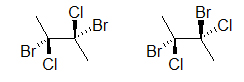
The two molecules above are different, prove this to yourself by edifice a model. An easier way to compare the two molecules is to rotate one of the bonds (here, it is the bond on the correct):
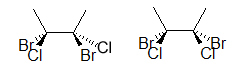
Notice how the molecule on the right has both bromines on the aforementioned side and chlorines on the same side, whereas the outset molecule is unlike. Read virtually dashed-wedged Line structures, lesser of folio, to understand what has been introduced higher up. You will larn more about the importance of atomic connectivity in molecules as yous continue on to learn about stereochemistry.
The following are examples of each, and how they can be used together.


Blue bead= OH group; White dewdrop=H
Higher up are four-carbon chains with attached OH groups or Cl and Br atoms. Remember that each line represents a bond and that the carbons and hydrogens accept been omitted. When you look at or draw these structures, the straight lines illustrate atoms and bonds that are in the same plane, the plane of the paper (in this case, computer screen). Dashed lines show atoms and bonds that go into the page, backside the plane, abroad from you. In the above example, the OH group is going into the plane, while at the aforementioned fourth dimension a hydrogen comes out (wedged).
Wedged lines illustrate bonds and atoms that come out of the page, in forepart of the aeroplane, toward you lot. In the second diagram above, the OH group is coming out of the plane of the paper, while a hydrogen goes in (dashed).

Blueish dewdrop= OH group; White dewdrop=H
Equally stated before, directly lines illustrate atoms and bonds that are in the same aeroplane equally the paper, just in the 2d instance, the straight line bond for OH ways that it it unsure or irrelevant whether OH is going abroad or toward y'all. It is also assumed that hydrogen is also connected to the aforementioned carbon that OH is on.

Blue dewdrop= OH group; H is not shown
Try using your model kit to see that the OH group cannot lie in the same plane at the carbon concatenation (don't forget your hydrogens!). In the final 2Dexample, both dashed and wedged lines are used because the attached atoms are not hydrogens (although dashed and wedged lines can be used for hydrogens).The chlorine is coming out the page while bromine is going into the folio.

Blueish dewdrop=Cl; Red dewdrop=Br
Source: https://chem.libretexts.org/Courses/SUNY_Potsdam/Book:_Organic_Chemistry_I_%28Walker%29/01:_Introduction/1.07:_3D_Structure
Posted by: markhamkinatim.blogspot.com


0 Response to "How To Draw Dash And Wedge Chemistry"
Post a Comment